-
Species of Origin
Human
Expression System
Escherichia coli
-
Affinity Tag
His Tag (C-term)
Storage Buffer
Lyophilized from a 0.2 µm filtered solution of PBS, pH 7.4.
-
Purity
>95% as determined by SDS-PAGE analysis.
Molecular weight
The protein has a calculated MW of 19.6 kDa.
The protein migrates as 20 kDa under reducing condition (SDS-PAGE analysis). -
Activity
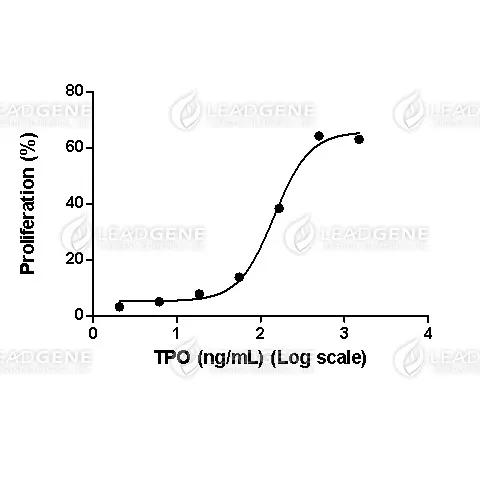
Measure by its ability to induce proliferation in MO7e cells. The ED₅₀ for this effect is <2 ng/mL.
Endotoxin Level
<0.05 EU per 1 µg of the protein by the LAL method.
-
Mycoplasma
Not detected
Form
Lyophilized
-
Specifications
-
Background
-
Background
Thrombopoietin (TPO) is a glycoprotein that produced by the liver, kidney, marrow stroma and several other tissues. The TPO level in the blood is mostly negatively correlated with the abundance of platelets and bone marrow megakaryocytes, although multiple states of inflammation or infection, liver failure, and hematological disturbances are associated with unexpectedly high or low circulating levels of the hormone.
Synonyms
Thrombopoietin, C-mpl ligand , ML, Megakaryocyte colony-stimulating factor, Megakaryocyte growth and development factor , MGDF, Myeloproliferative leukemia virus oncogene ligand
-
Uniprot ID
P40225
Sequence Note
Ser22-Leu195
-
-
Instruction
-
Reconstitution
It is recommended to reconstitute the lyophilized protein in sterile H₂O to a concentration not less than 0.5 mg/mL and incubate the stock solution for at least 20 min to ensure sufficient re-dissolved.
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at -20°C or lower for long term storage.
-
Stability & Storage
This product is stable after storage at:
- -20°C for 12 months in lyophilized state from date of receipt.
- -20°C or -80°C for 2 weeks under sterile conditions after reconstitution.
Avoid repeated freeze/thaw cycles.
Manufacturing Specifications
LeadGMP® recombinant proteins are manufactured in ISO 13485:2016 and GMP certified facility. The processes include:
- Animal-free reagent and laboratory
- Manufactured and tested under GMP guideline
- Testing and traceability of raw material
- Records of the maintenance and equipment calibration
- Personnel training records
- Batch-to-batch consistency
- Documentation of QA control and process changes
- Manufactured and tested under an ISO 13485:2016 certified quality management system
- Stability monitor of product shelf-life
-
-
Image
1/2 -
Review

Help others learn more about this product. Use the link below to share your experience.
-
Publication

There are currently no publications. Use the link below to let us know.
-
Datasheet & Documents
1/1
Disclaimer:For Research Use or Further Manufacturing Only.

