-
Species of Origin
Human
Expression System
Escherichia coli
-
Affinity Tag
His Tag (N-term)
Storage Buffer
Lyophilized from a 0.2 µm filtered solution of PBS, pH 8.0.
-
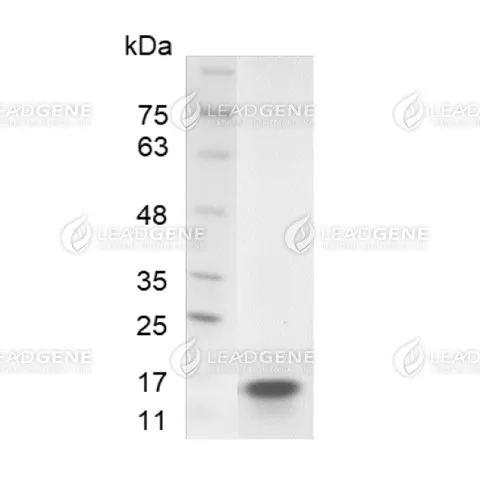
Purity
>98% as determined by SDS-PAGE analysis.
Molecular weight
The protein has a calculated MW of 15.4 kDa.
The protein migrates as 15 kDa under reducing condition (SDS-PAGE analysis). -
Activity
Measure by its ability to induce TF-1 cells proliferation. The ED₅₀ for this effect is <80 pg/mL. The specific activity of recombinant human GM-CSF is approximately >1 x 10⁷ IU/mg.
Endotoxin Level
<0.05 EU per 1 µg of the protein by the LAL method.
-
Mycoplasma
Not detected
Form
Lyophilized
-
Specifications
-
Background
-
Background
Granulocyte-macrophage colony-stimulating factor (GM-CSF) was first identified as a growth factor due to its ability to induce proliferation and differentiation of bone marrow progenitors into granulocytes and macrophages. GM-CSF is produced by multiple cell types including activated T cells, B cells, macrophages, endothelial cells and fibroblasts upon receiving immune stimuli. GM-CSF stimulates stem cells to produce granulocytes and monocytes functions as a cytokine.
Synonyms
Granulocyte-macrophage colony-stimulating factor, Colony-stimulating factor , CSF, Molgramostin, Sargramostim
-
Uniprot ID
P04141
Sequence Note
Ala18-Glu144
-
-
Instruction
-
Reconstitution
It is recommended to reconstitute the lyophilized protein in sterile H₂O to a concentration not less than 0.5 mg/mL and incubate the stock solution for at least 20 min to ensure sufficient re-dissolved.
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at -20°C or lower for long term storage.
-
Stability & Storage
This product is stable after storage at:
- -20°C for 12 months in lyophilized state from date of receipt.
- -20°C or -80°C for 1 month under sterile conditions after reconstitution.
Avoid repeated freeze/thaw cycles.
Manufacturing Specifications
LeadGMP® recombinant proteins are manufactured in ISO 13485:2016 and GMP certified facility. The processes include:
- Animal-free reagent and laboratory
- Manufactured and tested under GMP guideline
- Testing and traceability of raw material
- Records of the maintenance and equipment calibration
- Personnel training records
- Batch-to-batch consistency
- Documentation of QA control and process changes
- Manufactured and tested under an ISO 13485:2016 certified quality management system
- Stability monitor of product shelf-life
-
-
Image
1/1 -
Review

Help others learn more about this product. Use the link below to share your experience.
-
Publication

There are currently no publications. Use the link below to let us know.
-
Datasheet & Documents
1/1
Disclaimer:For Research Use or Further Manufacturing Only.
