-
Species of Origin
Human
Expression System
HEK293
-
Affinity Tag
His-SUMO Tag (N-term)
Storage Buffer
Lyophilized from a 0.2 µm filtered solution of PBS, pH 7.4.
-
Purity
>98% as determined by SDS-PAGE analysis.
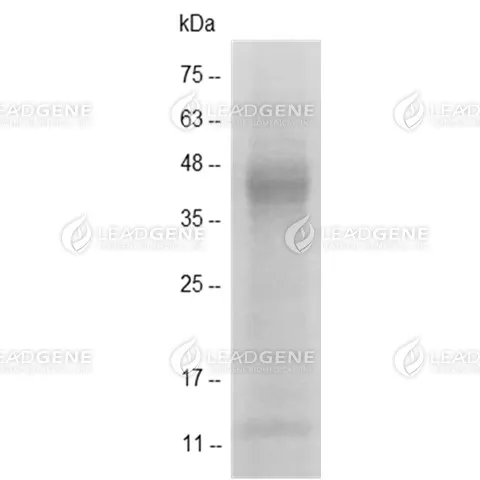
Molecular weight
The protein has a calculated MW of 36.42 kDa.
The protein migrates as 35-48 kDa under reducing condition (SDS-PAGE analysis). -
Activity
Measure by its ability to induce TNF alpha in RAW264.7 cells. The ED₅₀ for this effect is <10 μg/mL.
Endotoxin Level
<0.1 EU per 1 μg of the protein by the LAL method.
-
Form
Lyophilized
-
Specifications
-
Background
-
Background
High mobility group protein B1 protein (HMGB1) is the high mobility group box family of non-histone chromosomal proteins. Human HMGB1 is expressed as a 25 kDa single chain polypeptide containing three domains: two N-terminal HMG boxes A and B, and a negatively charged 30 a.a. C-terminal region that contains only Asp and Glu. Post-translational modification on HMGB1 have been reported, affects its localization, receptor interactions, and function. HMGB1, with a disulfide bond between C23 and C45 , have been reported that cause cytokine production and the activation of NF-κB. Otherwise, the fully oxidized form has no immune function, losing its proinflammatory effect and the apoptotic cell death activation function.
Synonyms
High mobility group protein B1, High mobility group protein 1, HMG-1
-
Uniprot ID
P09429
Sequence Note
Met1-Asp169
-
-
Instruction
-
Reconstitution
It is recommended to reconstitute the lyophilized protein in sterile H₂O to a concentration of 200 μg/mL and incubate the stock solution for at least 20 min to ensure sufficient re-dissolved.
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at -20°C or lower for long term storage.
-
Stability & Storage
This product is stable after storage at:
- -20°C for 12 months in lyophilized state from date of receipt.
- -20°C or -80°C for 1 month under sterile conditions after reconstitution.
Avoid repeated freeze/thaw cycles.
-
-
Image
1/1 -
Review

Help others learn more about this product. Use the link below to share your experience.
-
Publication

There are currently no publications. Use the link below to let us know.
-
Datasheet & Documents
1/1
Disclaimer:For Research Use or Further Manufacturing Only.
